Making molecular sieves and filters more efficient and safer is now possible thanks to the use of X-rays. The study has just been published in Nature Communications by a team of researchers from the Politecnico di Milano, the Grenoble Synchrotron and the University of Heidelberg.
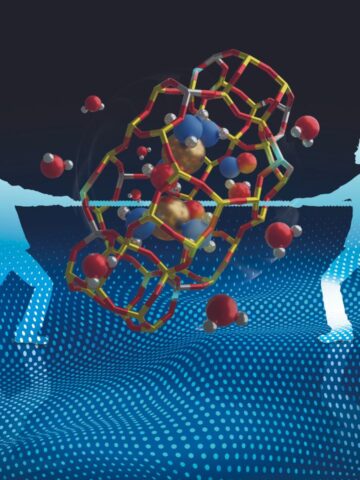
Nanometric filters are tools with which we can capture unwanted, even very small molecules such as CO2, other polluting gases and hazardous volatile organic substances. In the last few years sieves based on a metal-organic framework (MOFs), equipped with nanopores of varying sizes, shapes and characteristics that can be adapted to the type of molecule that you want to capture, are becoming more widespread.
Nowadays, the production of MOFs is limitless, but only a few have the ideal characteristics for efficient filtration. It is fundamental to be able to determine, for example, if only the desired molecules remain trapped in the pores and the efficiency of their sequestration. The trapped molecules, in fact, do not sort easily in the interstices and are difficult to see by X-ray diffraction, which often causes interpreting errors by overestimating or underestimating the efficacy of a certain material.
With this work, the methodology to be followed in order to locate molecules disorderly trapped in the interstices of the MOFs has become clear. In addition to a good quality of the crystalline matrix of the MOF (before and after its use), an accurate diffraction analysis is required. With our proposed protocol, the nanoporous materials can be analysed more accurately and their efficacy in filtration can be better established. Following the suggested procedure, a group of researchers from the Beijing Polytechnic University have been able to re-analyse several previous results concerning a MOF capable of trapping dioxin molecules. This is just one example of the possible applications in the field of materials engineering.
Piero Macchi, professor of the Department of Chemistry, Materials and Chemical Engineering “Giulio Natta”
Recent studies have demonstrated how the MOFs could be used, for example, in the reduction of CO2 in industrial exhaust gases, or as a water resource in highly dry and desert areas for their extraordinary capacity to absorb water from the atmosphere, even those with low humidity. For these and other uses, it is fundamental to determine the true capacity of the material to absorb the desired molecules.