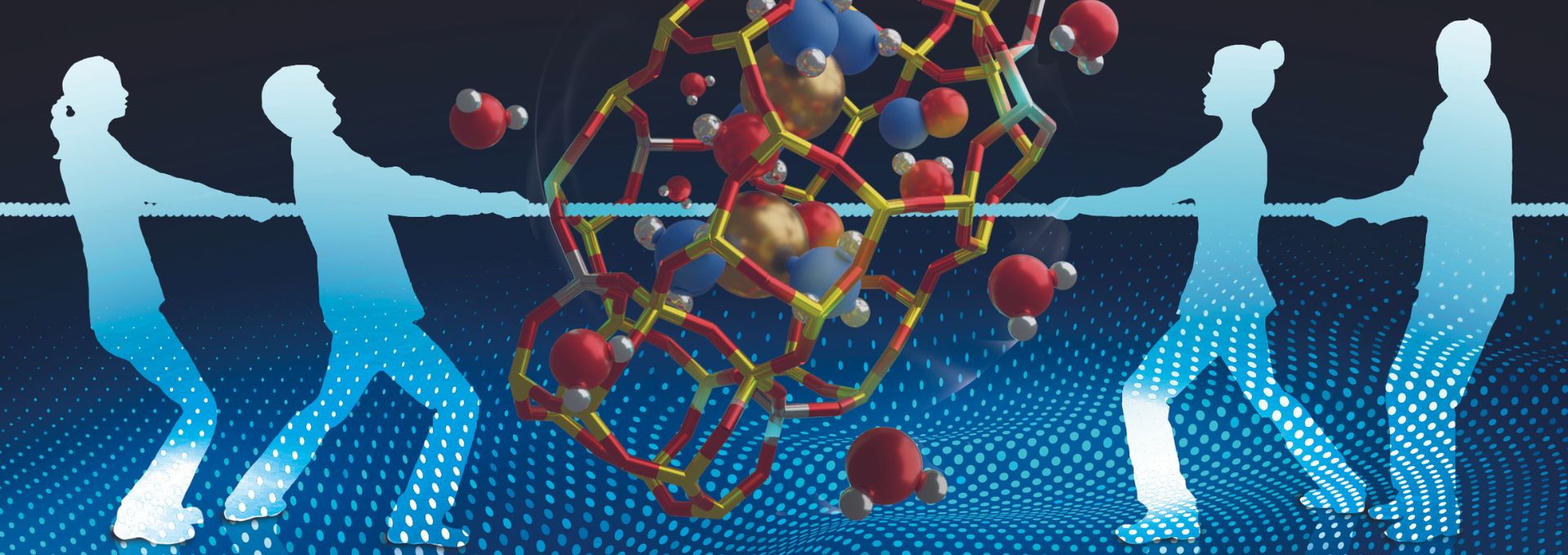
A study conducted by the Department of Energy‘s ‘Laboratory of Catalysis and Catalytic Processes (LCCP)’ group at Politecnico di Milano has been selected as the front cover of the journal ChemCatChem. The article highlights the importance of describing the complex catalytic phenomena involved in pollutant (NOx) abatement reactions from a molecular point of view.
For the first time ever, this study offers a theoretical understanding of the role of water in the NH3-SCR process: this is technologically relevant in the field of pollutant abatement because H2O is abundantly present as a reaction product.
Enrico Tronconi, co-author of the study
The study shows that the presence of water in the reduction loop of the NH3-SCR redox mechanism changes the reaction environment at the molecular level. On the one hand, the addition of water in the system leads to a lowering of the activation energy of the reaction due to kinetically relevant enthalpy stabilisation effects of the transition state. On the other hand, the reduced mobility of the transition state leads to entropic penalties that lower the reaction speed.
Our research explores the engineering significance of an atomistic study of catalytic mechanisms, providing a detailed description of the elementary steps and molecular identities involved in reactions. These tools, in synergy with experimental work, play a major role in the optimisation and development of catalytic processes.
Matteo Maestri, co-author of the study